Desmoid tumor (DT), also known as aggressive fibromatosis, is a rare disease characterized by fibroblastic proliferation arising from connective tissue of the muscles, fascia, or aponeurosis. This entity represents 0.03% of all neoplasms and 3% of all soft tissue tumors.1 Although histologically benign, DTs are locally infiltrative with a recurrence rate of up to 30% in variable times after surgical excision. According to their anatomic location DTs are classified into: intra- abdominal, abdominal wall, and extra-abdominal wall. Head and neck DTs are one of the extra-abdominal DTs and estimated to constitute only 7%–15% of all DTs throughout the body.2 In the literature, reports on head and neck desmoid tumors were few and most of them only had a small number of cases.3–5
DTs usually affect females, and the most affected age range is 30–40 years. The etiopathogenesis of DT is unknown, however some cases have been related to prior trauma or surgery, and more rarely with adenomatous polyposis coli (APC) germline mutations.
Treatment of these tumors may require a multidisciplinary approach. Surgery contraindication may be due to tumor size or location, which could lead to serious functional impairment. Systemic severe disease may also be a surgery contraindication. In these cases, treatment can also include primary radiotherapy and systemic approaches, including cytotoxic chemotherapy, administration of imatinib, nonsteroidal anti-inflammatory drugs, and hormonal therapy. Otherwise, a “wait-and-see” policy is an option for asymptomatic cases.1
The aim of this study is to present the case report of a patient with a giant DT of the neck related to a self-injury event years before; the mass measured 20 × 22 cm, and no case with this measurement has been previously published. We report the clinical evaluation, the results of image diagnosis, the surgical procedure, and the followup.
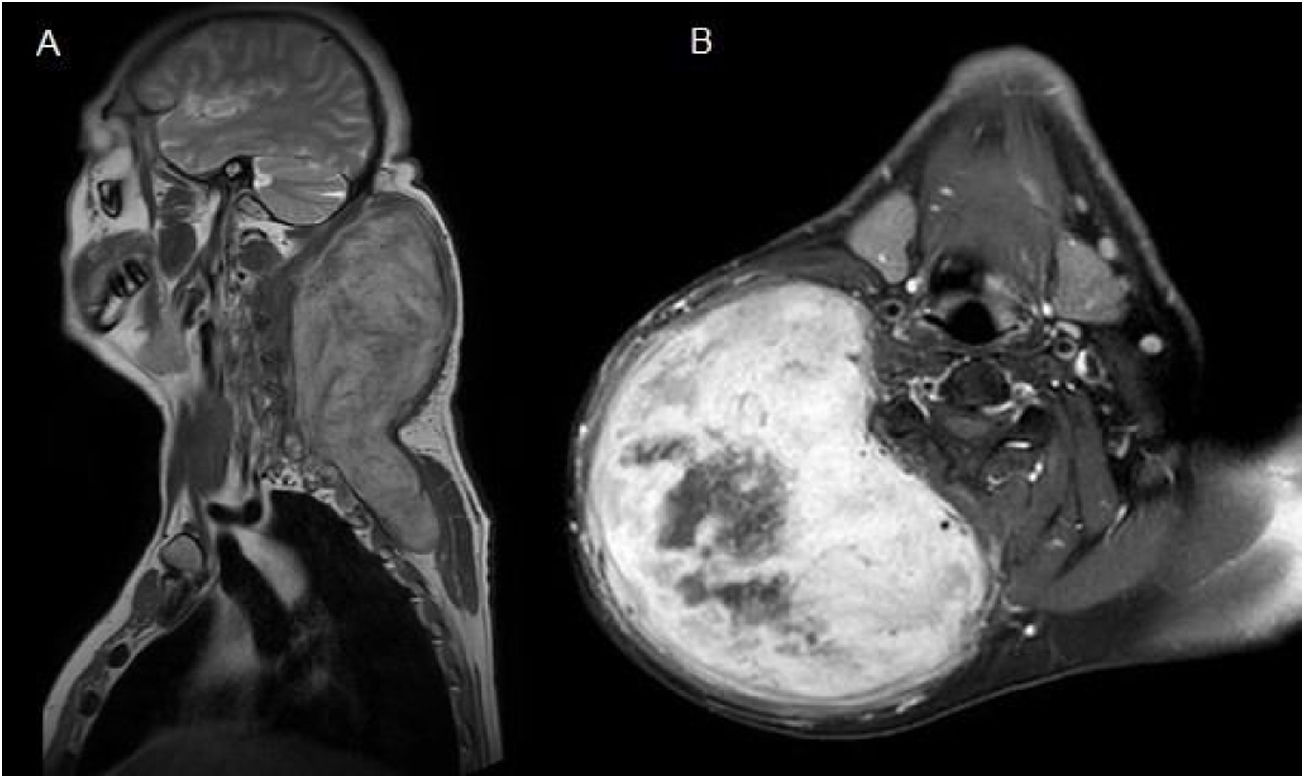
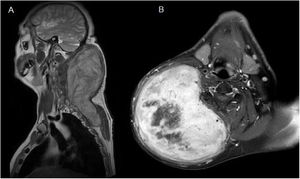
Case reportA 52-year-old man presented with a huge and firm mass in the posterior right lateral area of the neck with associated restriction of movement. The patient had background of a self-inflicted injury with a knife 4 years prior. He had no other significant medical history. The magnetic resonance imaging (MRI) showed the presence of a subcutaneous mass measuring 13 × 14 × 22 cm in the right posterior lateral area of the neck extending into the posterior thoracic wall. The images were hypointense in T1-weighted images and hyperintense in T2-weighted images. Furthermore, the T1-weighted images showed significant enhancement of the mass and highlighted a central necrotic area (Fig. 1). Fine- needle aspiration was performed. The mass was interpreted as a spindle-shaped cell tumor without evidence of atypia, consistent with fibromatosis.
(A) Coronal T1-weighted Magnetic Resonance Imaging (MRI) showed the presence of a subcutaneous mass measuring 13 × 14 × 22 cm in the posterior area of the neck which extended into the posterior thoracic wall. (B) Axial T1-weighted MRI showed significant enhancement of the mass and highlighted a central necrotic area.
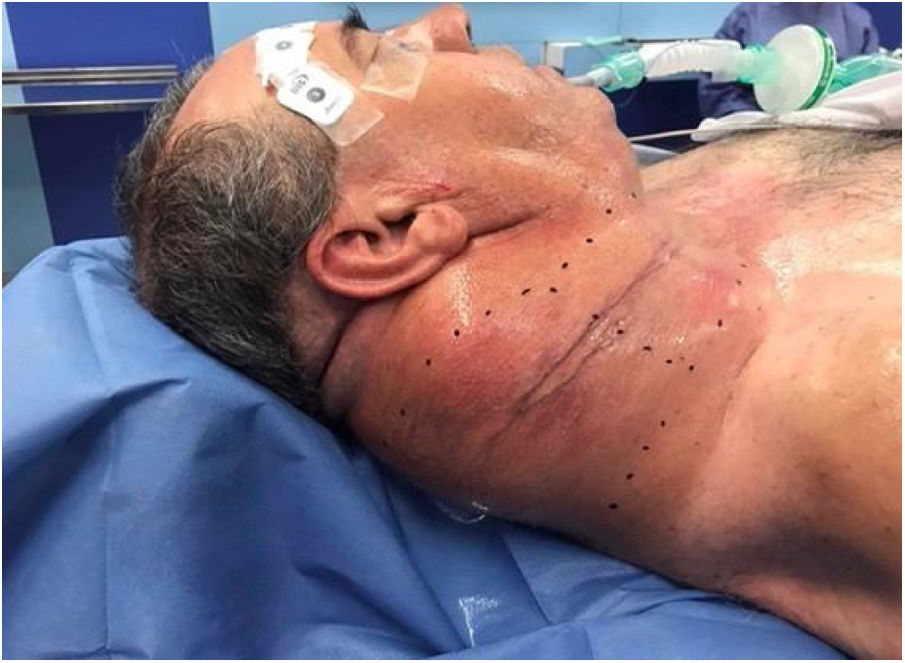
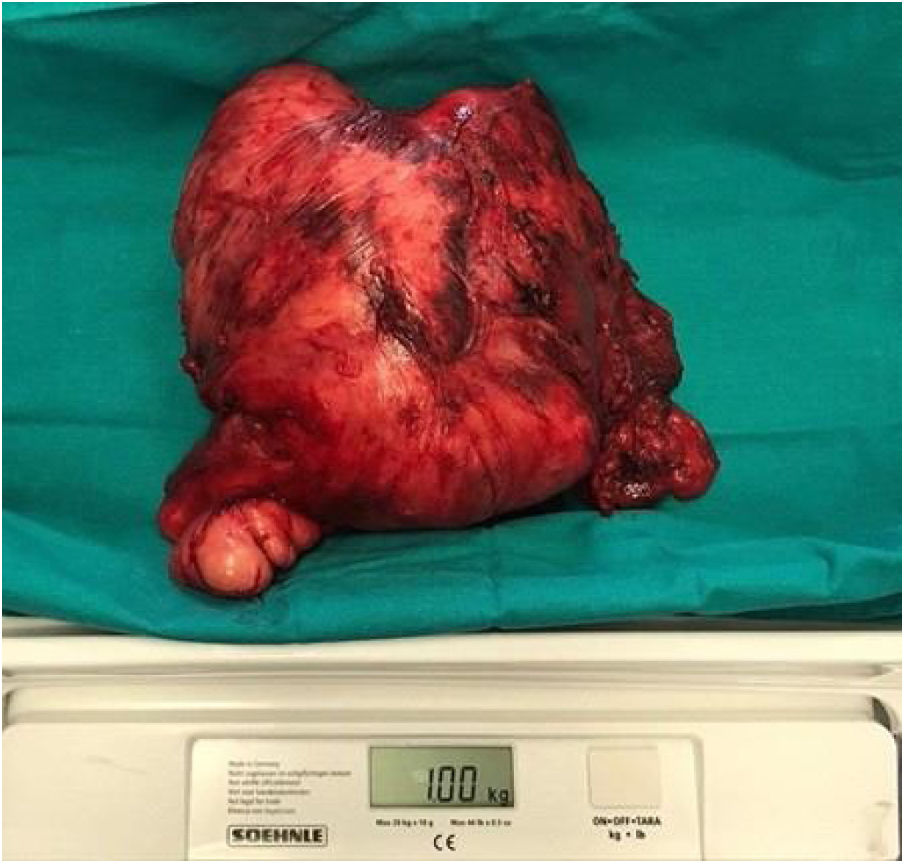
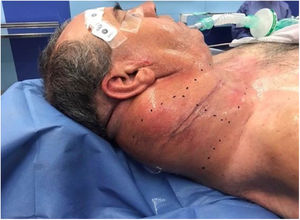
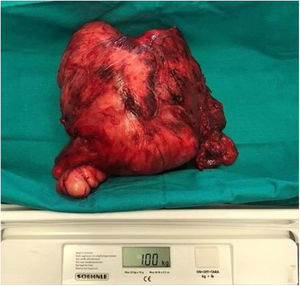
Following discussion by the multidisciplinary oncological group surgical treatment was elected. Preoperative planning was carried out to decide the best surgical approach to resect the cervical giant mass. We decided to used a Mayfield® headrest to provide access to the posterior neck area, employing a Hayes-Martin incision to approach the infra-auricular and supraclavicular area (Fig. 2). The dermoplatysmal flap was raised in the anterior part, the sternocleidomastoid muscle and vascular axis were identified, the tumor was dissected from the splenius and scalene muscles. During the operation, the tumor was noted to be in tight adhesion to and invading the adjacent fascia and musculature, especially the trapezius musculature. The tumor boundaries were limited by the base of the skull, inferiorly by the lower third of the scapula, and medially by the styloid process which was sectioned with the Riolano bunch. Complete excision of the tumor was accomplished: it measured 20 × 22 × 5 cm, the macroscopic appearance was of a rounded, firm, and well-circumscribed mass (Fig. 3). A cross-section cut revealed a white and whorled surface resembling scar tissue and focal edematous and cystic alterations, but no evidence of necrosis. The intraoperative pathological diagnosis confirmed the benign character of the tumor. No additional neck dissection was performed.
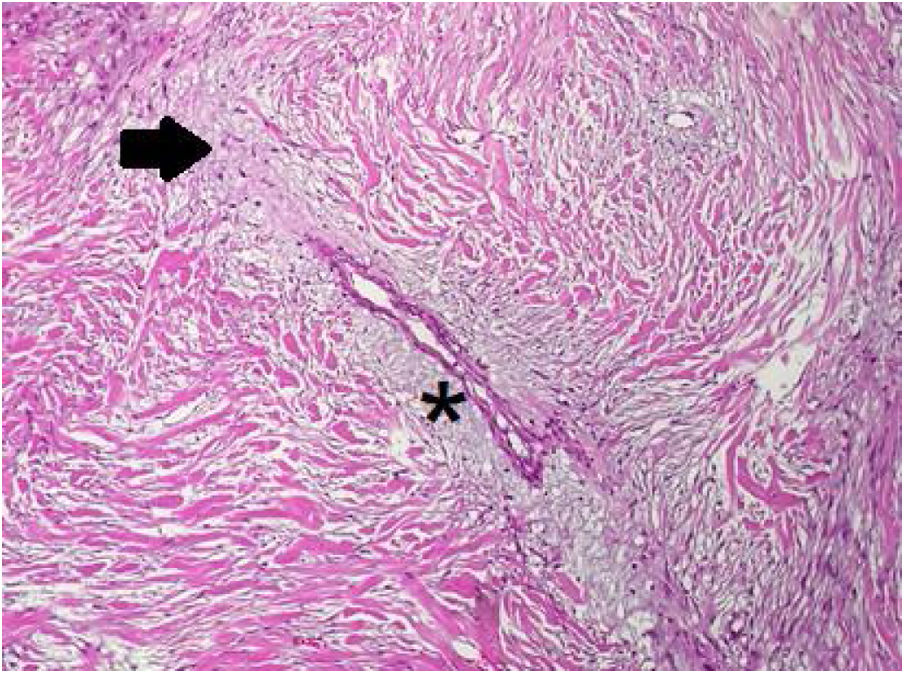
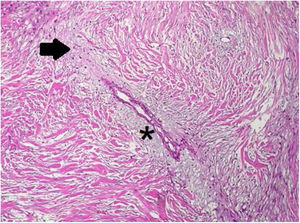
In the pathology examination, the lesion comprised spindle-shaped cells arranged in long fascicles. Some sections were mostly cellular, while others were markedly collagenous with keloidal hyalinization (Fig. 4). Characteristic regenerative skeletal muscle fibers were entrapped toward the edges of the tumor. The macroscopically described edematous regions correlated with myxoid alterations and ischemic necrosis.
Postoperative radiotherapy was dismissed because the surgical margins were tumor-free when assessed by histopathological examination. Functional sequela in elevation and dorsal movement of the shoulder due to partial resection of the trapezius muscle was detected in the patient after the surgery. No recurrent lesion was observed at his sixth month postoperative MRI follow-up examination.
DiscussionDT of head and neck is a rare neoplasm that, even though it has no metastatic potential, requires accurate microscopic interpretation, and represents a significant therapeutic challenge due to the unique anatomy of the head and neck region. Soft tissue spindle cell tumors to have diagnostic challenges for pathologists. In this clinical case, the morphological features of the case raised suspicion of the presence of a low-grade sarcoma, such as myxofibrosarcoma or a malignant peripheral nerve sheath tumor. However, the lack of nuclear atypia and the expression of smooth muscle actin, as well as negative staining for EMA, MUC4, and S100, allowed us to discard these malignant neoplasm diagnoses. Only by gathering all the data, and putting a special emphasis on the clinical context, were we able to make a final clinical interpretation of fibromatosis.
Similar to other benign tumors of the head and neck, wide surgical resection with clear margins is the most common first-line treatment for DT. However, DT tends to be associated with significant morbidity when it occurs in the head and neck region, due to the infiltration or proximity to vital anatomic structures and a tendency for local recurrence. Sometimes complete resection of these tumors is difficult and although the margin status is of importance, operations that preserve function and structure should be the primary goal.1
Although the reported recurrence rate is high with a range between 46% and 62%,6 the adjuvant management (suggested for adequate local control) has not been established because the incidence of head and neck DT is too rare to conduct a comprehensive study to establish the role of adjuvant therapy. Moreover, spontaneous regression and arrested growth after incomplete resection are occasionally observed, and the section margin is poorly correlated with the development of local recurrence.7
Adjuvant treatment options include radiotherapy; a “wait-and-see policy”; and medical treatment (e.g., nonsteroidal anti-inflammatory drugs, hormonal therapy, chemotherapy or imatinib). When the tumor behavior is not progressive, observation may be the most appropriate management in asymptomatic patients. Radiotherapy is considerably effective in local tumor control (not equivalent to tumor eradication) but is associated with a high rate of complications that are dose- dependent.8 Radiotherapy should only be applied where the total excision would lead to mutilation and radiation-induced morbidity is expected to be acceptable.1
Recurrences in the head and neck generally arise in the first 2-years, but they may occur anytime from a few months to >10 years after the initial surgery.2 For disease extension investigation and followup examinations, MRI is the imaging modality of choice because it provides better soft tissue definition than computed tomography.9 Given the unknown natural behavior of these tumors, patients require close lifelong monitoring.
ConclusionDesmoid is a rare and heterogeneous disease, which definitely requires individualized treatment to reduce the local tumor control failure. We performed a complete surgical resection with clear margins in this case: it was challenging due the size of the tumor and the location. After searching in PubMed index no case of such a DT in the head and neck region has been previously published. A period of careful followup is indicated to assess the behavior of the tumor.
Conflicts of interestThe authors declare no conflicts of interest.
Peer Review under the responsibility of Associação Brasileira de Otorrinolaringologia e Cirurgia Cérvico-Facial.